One week into an internship at NASA's Jet Propulsion Laboratory (JPL)
I am happy to say that I have completed my first week of my internship at JPL! The experience has been tremendously positive so far. JPL is a truly amazing place. The JPL campus consists of approximately 200 different buildings that make up a small “city of scientists and engineers” (they even have their own fire department!), situated at the southern edge of the San Gabriel mountains near Pasadena, California. Here’s a view of the campus from about a mile or two away:
JPL, a city of science on a hill. Photo taken by yours truly.
JPL is a federally-funded research and development center (FFRDC), owned by NASA and managed by California Institute of Technology (Caltech). At any given time, there are six to seven thousand engineers and scientists working here, the majority collaborating on projects related to space exploration. JPL has played a pivotal role in some of the most successful space missions to date, such as the Voyager missions, the Galileo and Cassini missions, and the Mars Science Laboratory (Curiosity rover), just to name a few. If you’d like to see more information about what is currently going on at JPL or what they have accomplished in the past, head on over to their main website, JPL.Nasa.gov.
The atmosphere at JPL is so wonderful. People are there to work on amazing science, and most seem truly thrilled to be doing just that. It’s hard not to be smiling just walking around JPL, because everyone else is! Many parts of the campus are quite beautiful, and some are even scenic, with decent views of the nearby mountains. There are a lot of outdoor spots for lunch, meetings and collaboration, or just grabbing a coffee. There are even several ‘coffee carts’ that serve Starbucks coffee, which is great, because, as my Calculus 1 professor used to say,
Coffee is the lubricant on the gears of science.
—Mark Beintema
“The Mall” at JPL, a popular spot for lunch, coffee, and enjoying the California weather. Photo taken by me.
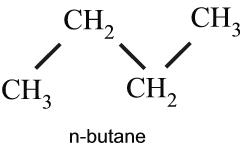
I will be working in the field of molecular spectroscopy this summer under my mentor, Dr. Keeyoon Sung. We will be measuring the infrared cross sections (explanation to follow) of the n-butane (C4H10) molecule in a variety of temperatures and pressures that are chosen to model the stratosphere of Saturn’s extremely interesting moon, Titan.
Structure of n-butane, a zig-zag chain of carbon atoms, each with a pair or triplet of hydrogen atoms.
Below, you can see an artist’s rendition of the view from the surface of Titan. Hydrocarbons, primarily methane and ethane, play a similar role on Titan as water does here on Earth. A methane lake is seen in this particular image.
Image credit: Original artist unknown, in spite of my best effort to identify him or her…
The goal of my project is to measure the cross section of n-butane as a function of wavelength. The cross section of a molecule is effectively how “big” the molecule is, with respect to incoming radiation. In other words, the larger the cross section of a molecule, the larger the chance that it will “catch” and absorb an incoming photon (particle of light). And again, this is all wavelength (or energy) dependent. The smaller the wavelength of a photon, the larger the energy, and every atom or molecule is very good at absorbing photons of specific energies, and not so good at absorbing photons of other energies. This all arises from the quantum mechanical nature of the universe. Take a look at the spectrum of n-butane below.
The spectrum of n-butane. Image Source: The National Institute of Standards and Technology (NIST) - Chemistry WebBook
If you’ve never seen one of these spectra before, take a moment to understand it, it’s quite straightforward. On the x-axis, we have the wavelength of light. Light is more and more energetic the smaller and smaller the wavelength gets, so on the plot above, the energy of the light being considered decreases as you move further and further to the right. On the y-axis, we are plotting how much of the light of that wavelength, when directed at n-butane, is transmitted, rather than absorbed. A transmittance of 1.0 means full transmission: the light of that particular wavelength passes right through the n-butane, every time. We might say that the molecule is “transparent” to that kind of light. Now, note the sudden dips in the plot at key wavelengths, like the one at wavelength of approximately 3.6 micrometers. It turns out that n-butane is highly absorbing at this (and some other) wavelengths. These wavelengths correspond to specific amounts of energy that n-butane is capable of accepting, so to speak. So, if I aim a photon with wavelength of 3.6 micrometers at a n-butane molecule, there’s a very high chance that the photon will be absorbed by the molecule, “exciting it” and causing some part of it to vibrate and possibly rotate. The type of excitation (i.e. perhaps one of the carbon-carbon bonds begins to stretch in and out repeatedly, or perhaps one of the methyl (CH3) groups on the edge of the molecule begins to rotate) depends on the energy of the absorbed light. The kicker to all this is that every atom and molecule has a unique spectrum, a unique “signature”, of sorts. The form of this signature is governed by quantum mechanics. Through quantum mechanics we can predict the spectra of molecules, and through experimental spectroscopy, we can measure and understand these signatures empirically. Capitalizing on both of these approaches allows us to “decipher” the signature of a molecule to high-precision in the lab, and this allows us to identify the presence of that molecule elsewhere in the universe by simply paying attention to what light is being emitted there.
Understanding the energy-dependent cross sections of n-butane (and other hydrocarbons) is crucial toward making sense of the data that the Cassini mission (and others, like Voyager) have collected from the atmosphere of Titan. These data hint at the presence of butane and other interesting hydrocarbons, but without extremely high-resolution measurements of how these molecules emit radiation in a controlled laboratory setting, it’s difficult to definitively identify them within the Titan atmosphere (and elsewhere). My task will be to analyze the spectrum of butane, calculate the cross sections as functions of wavelength, temperature, and pressure, and publish these results so that astronomers working with the Cassini data to model the Titan atmosphere have a better foundation on which to work.
So, why does knowing the precise details of Titan’s atmospheric makeup really matter? Here are two interesting applications to this sort of work. The first is that Titan, in its present form, may be similar to the Earth back in its early development, long before life evolved (see Clark & Ferris, Chemical evolution on Titan: comparisons to the prebiotic Earth, in Origins of Life and Evolution of Biospheres, 1997). Thus, from a planetary science perspective, understanding the components of Titan’s current atmosphere can inform our understanding of the early development of the Earth as a planetary system. And further, from a biological perspective, understanding Titan’s atmosphere may even shed light on the development of life itself! (see Trainer et al., Organic haze on Titan and the early Earth, Proceedings of the National Academy of Sciences of the United States of America, 2006)
The second application of my work is with regards to future Titan missions. When you land a spacecraft on a planet/moon with an atmosphere, it is crucial to know everything you can about the atmosphere: temperature and pressure profiles, density profiles of each molecular constituent, etc. This allows engineers to make informed decisions when it comes to designing the landing stage, particularly the heat shield and parachutes for the lander, which are crucial in achieving a safe landing. We’ve already landed on Titan once (see image below from the Cassini-Huygens probe), and chances are, we’ll go back again sometime soon!
The surface of Titan as seen by the Huygens probe in 2005.
Anyway, I feel like I have an amazing opportunity on my hands here working at JPL this summer. My plan is to continue to document my experience here on this blog for anyone interested in my work and how I’m spending my time during this summer. My intention is to update once a week, and I’ll do my best to stick to that, most likely updating each weekend. I hope you’ll check back occasionally for more!
Have a question related to this post, or just want to get in touch? Feel free to Contact me.